
At each visit, several outcomes including the area, ulcer closure, and granulation tissue formation were assessed. ( 4) patients were examined weekly for the first 4 weeks and then every 2 weeks until the end of the study (16 weeks) or ulcer closure. ( 6) the sponsor prepared the randomization scheme and were distributed in sealed envelopes containing the treatment assignment to be opened sequentially as patients were enrolled. At patient randomization, treatment was assigned on the basis of the next sequentially labelled envelope. Envelopes were sequentially numbered before site distribution. Numbers were assigned to a treatment group and then sealed in opaque envelopes containing black paper labelled with treatment and patient ID. ( 4) randomization was accomplished by generating blocks of numbers. In these trials, it was not possible to mask the investigators or the patients to the treatment assignment. In both studies data was analyzed based on intention to treat principle. Both studies reported 80% power for detection of 20% between group differences in the proportion of patients who achieve the primary outcome. The primary outcome and its definition were similar in the two studies. This study enrolled 162 patients with a mean age of 59 years. ( 6) investigated the effectiveness of NPWT in the treatment of amputation wounds of the foot in patients with diabetes and compared the effectiveness of the NPWT with moist wound therapy. The study enrolled 342 patients with a mean age of 58 years. ( 4) investigated the effectiveness of NPWT in the treatment of diabetic foot ulcers compared with advanced moist wound therapy. Both trials were multicentre and the study period was 16 weeks in both. 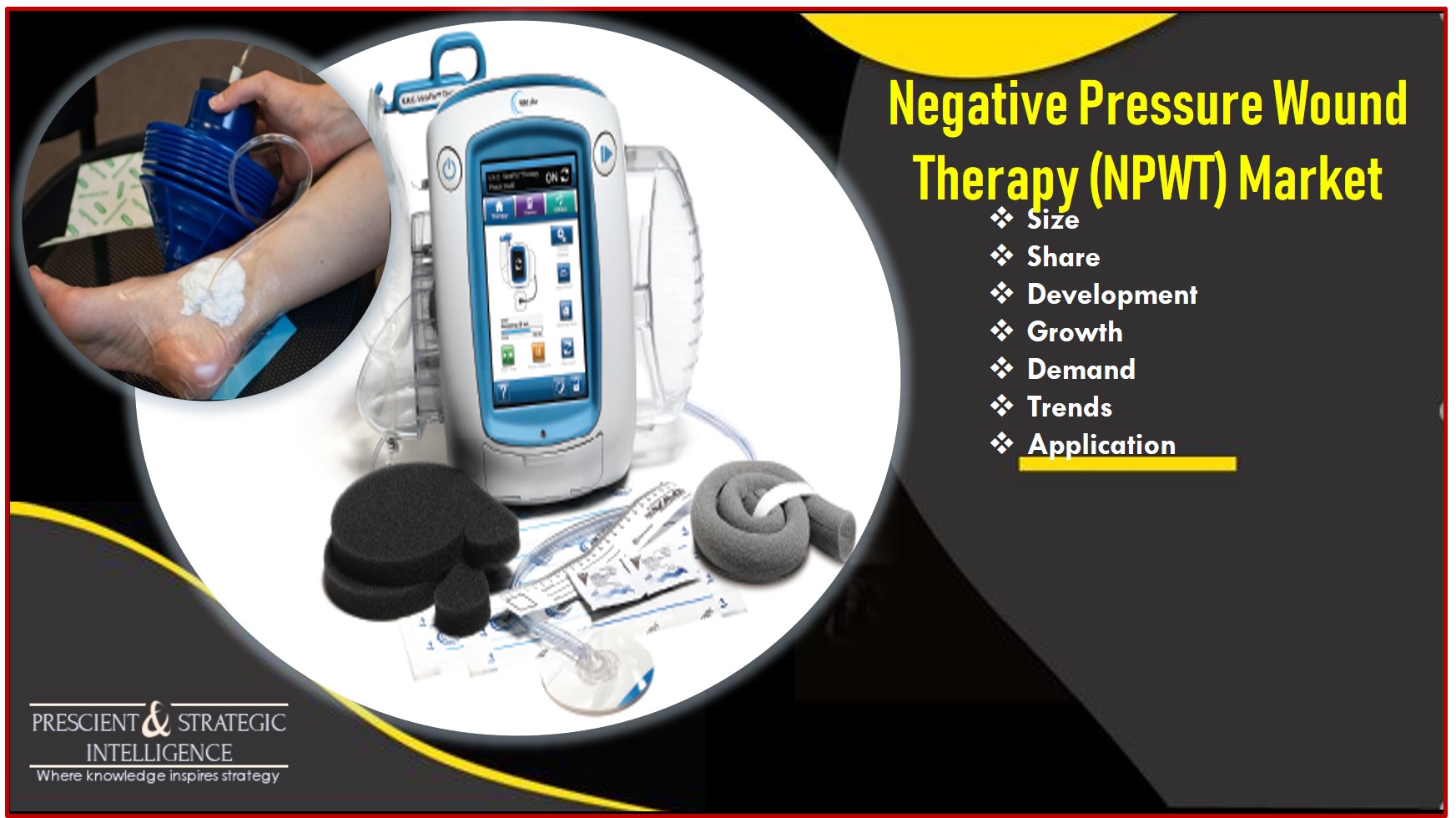
The two trials on diabetic foot ulcer ( 4 6) investigated the NPWT system manufactured by KCI and both received research funding from the manufacturer. All of the systematic reviews noted that there was a lack of high-quality evidence to support NPWT versus other treatments. While the authors were able to capture data on adverse events, however, due to the lack of studies comparing one NPWT system to another NPWT system, they were unable to determine the severity of adverse events for one NPWT system compared to another.Īs part of their systematic review, the AHRQ identified 22 existing systematic reviews (2000-2008) that included studies on NPWT versus other wound treatments for patients with a broad range of wound types. The AHRQ review concluded that “at this time the available evidence cannot be used to determine a significant therapeutic distinction of a NPWT system”. Nineteen studies reported fewer complications in patients receiving NPWT compared to standard care while eight studies reported that the two groups had similar adverse events. Seven studies described NPWT as a safe treatment. 
Thus, any quantitative synthesis of the data was not possible.Īdverse events were reported in 37 of the 40 studies comparing NPWT to other treatments. Studies included in the AHRQ review were very heterogeneous in regards to the patient population and wound type, primary outcomes, and method of outcome measurement. 
Two studies investigated the effectiveness of NPWT in skin grafts. The majority of studies examined chronic wounds. The sample sizes ranged from 22 patients to 342 patients. Of the 40 studies included in the AHRQ review, 9 studies were RCTs. The primary objective of the AHRQ review was to examine the comparative effectiveness of different NPWT systems and their components as well as the complication rates of different NPWT systems. A systematic review by the Agency for Healthcare Research and Quality (AHRQ) ( 3) included published studies from 2000 to 2008. The search identified 247 citations including several existing systematic reviews and health technology assessments.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
